Introduction: The Compounding Shift in 2025
The story of semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, is an intriguing tale of success, drug shortage and unintentional consequences. It was first used to treat type 2 diabetes and then for weight loss, and is very effective, leading to a surge in demand.
The US Food and Drug Administration (FDA) used its enforcement discretion policy to allow state-licensed compounding pharmacies and registered outsourcing facilities to compound semaglutide. The policy is outlined in its guidance on compounding during an active drug shortage which explains that only FDA-approved drug substances can be used and all quality and safety requirements must be met.
This has been reflected in the medical literature. Pharmacoepidemiologists and endocrinologists have raised concern that the rapid increase in prescriptions for GLP-1s has, in some cases, not kept up with clinical and regulatory pathways to ensure safety and efficacy.
Let’s look at these factors in detail to offer an evidence-based perspective on why compounded semaglutide may not be effective and what patients can do to seek safe and effective treatment.
1. The Root of the “Salt” vs “Base” Problem of Active Ingredient Purity
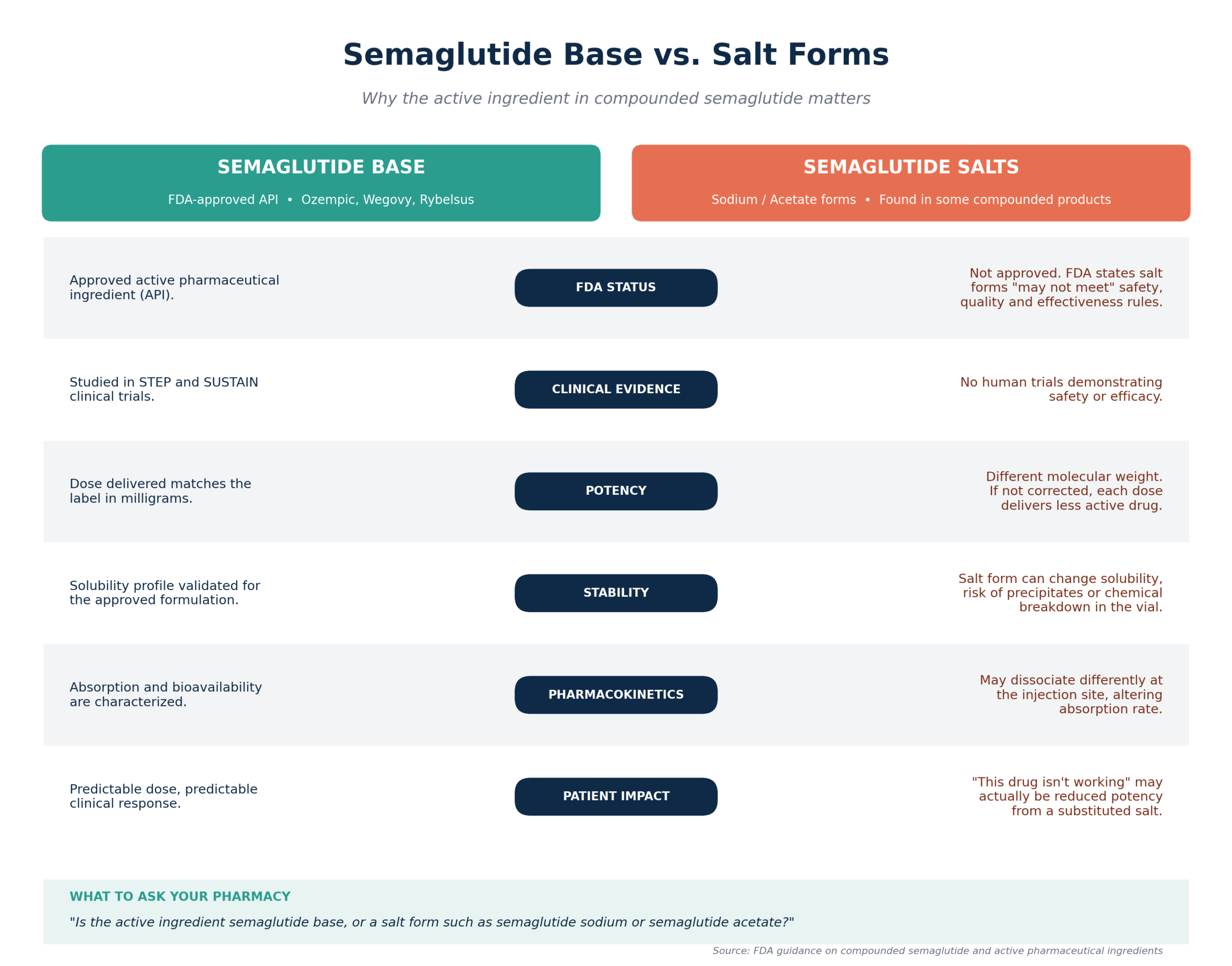
The most obvious factor for drug efficacy is the structure of the active pharmaceutical ingredient (API). For the FDA-approved semaglutide (brand names Ozempic, Wegovy and Rybelsus), the API is semaglutide base. The molecular structure (long chain peptide with a fatty acid side chain) was used in the STEP and SUSTAIN trials to demonstrate the safety and efficacy of the drug.
However, the innovator has a worldwide API supply of semaglutide base. But some chemical suppliers can offer more readily available compounds such as semaglutide sodium and semaglutide acetate. These are peptides salts. This is a significant difference.
The FDA Warning on Compounded Semaglutide has publicly acknowledged that some products are using these salts which are not the same as the active pharmaceutical ingredient (API). The FDA indicates that products that are labelled as semaglutide, but are containing these salts “may not meet the FDA’s requirements for safety, quality and effectiveness”.
This statement is founded on good scientific and regulatory grounds. The FDA’s general guidance on active pharmaceutical ingredients notes that a minor change in the salt form, ester or complex of a drug substance may result in significant changes in its properties. These differences can affect critical parameters:
- Solubility and Stability: Salt form may affect solubility in the vehicle used to compound the drug substance (usually bacteriostatic water), which can affect the clarity and stability of the solution. Precipitates or breaks down during the shelf life of the vial, which could affect dose.
- Pharmacokinetics: How the drug is absorbed and the potency (the amount of drug that reaches the bloodstream) may be different. For instance, a salt could dissociate differently at the injection site, which may lead to changes in absorption rate compared to the drug used during the original drug development process.
- Potency: The molecular weight of the sodium form of semaglutide is different from the base. If a pharmacy compounds a vial to deliver a particular dose (in milligrams) of semaglutide base, but uses the sodium salt without taking into account the difference in molecular weight, then less active molecule will be delivered with each dose.
The official United States Pharmacopeia (USP), the book that sets the standards for medicines in the U.S. stresses that these types of peptide and biologic drugs are particularly sensitive to formulation. These monographs and general chapters guarantee the identity, strength, quality and purity of these large and complex molecules are maintained from manufacturing to administration.
Impact on Patients: A patient receiving a vial of semaglutide sodium compounded may be receiving a product with changed, likely reduced, potency. The feeling of “this drug is not as good as it should be” could be due to this substitution, and not obvious to the patient.
2. The Effects of Cold Chain Failures on Potency Loss in a Fragile Molecular Structure
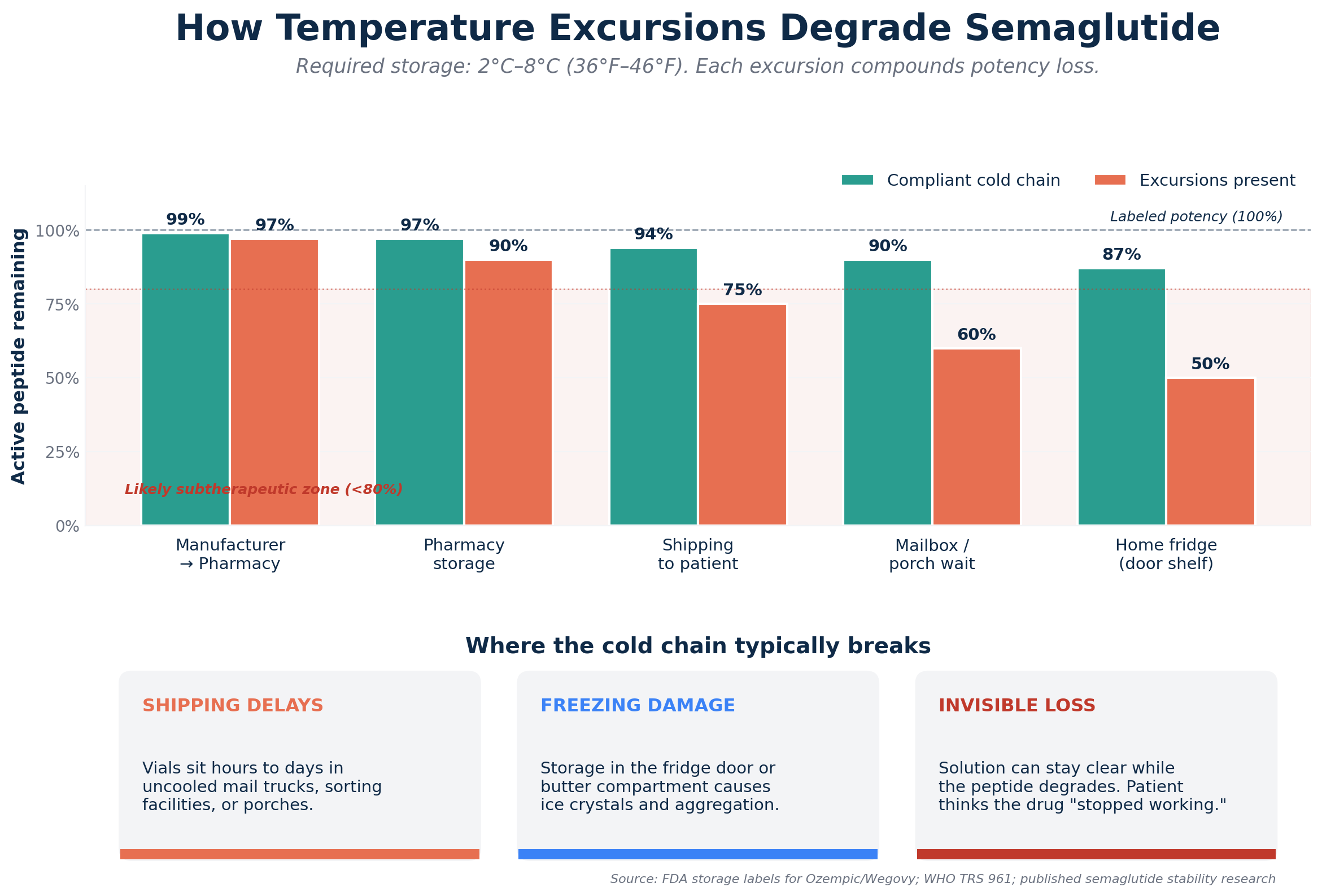
Semaglutide is not a hardy small molecule such as aspirin. It’s a large, intricate peptide and the molecule’s structure is critical for it to work. This shape is maintained by tethers that are susceptible to temperature.
FDA labels for Ozempic and Wegovy require that these products be stored at 2°C to 8°C (36°F to 46°F) until the end of their shelf life and for 56 days (or as indicated) after they have been opened. This is not a recommendation, but a requirement, based on stability studies.
FDA Drug Storage and Handling emphasises the need for cold chains in biologics and other temperature sensitive medicines. Excursions (deviations) from required conditions can cause “irreversible degradation” (physical such as aggregation and precipitation or chemical such as deamidation and oxidation) rendering the drug ineffective or inoperative.
The WHO Technical Report Series 961 on stability of biotherapeutic products calls peptides and proteins the most sensitive of drugs. Higher temperatures increase the activity of molecules and can lead to degradation and lower temperatures can cause ice crystals to form and phases to separate.
This is supported by the science of pharmaceuticals. Research of the impact of buffer capacity, pH and temperature on the stability of semaglutide have meticulously described how excursions at different temperatures produce increases in the degradation products and reductions in the amount of intact and active peptide in solution.
This can happen without the solution becoming cloudy. The impact on the patient receiving semaglutide compounded for them is multi-fold:
- Shipping: Compounding pharmacies often ship drugs. The drug may be waiting in the mailbox, non-temperature-controlled vehicle or sorting facility for hours to days.
- Pharmacy: Is there a rigorous cold chain from receipt of API to shipment?
- Home storage: Is it stored in the body, not the door or butter compartment (where it would freeze) of the refrigerator?
The efficacy of the vial can be compromised by 20%, 50% or more due to one large temperature excursion. The patient’s dose is subtherapeutic and they don’t get better and are disappointed.
3. Vial Misuse and the difference between beyond-use and expiration dates
Potency is influenced by this common source of confusion. The expiration date is determined by a manufacturer after long-term, real-time stability studies under controlled conditions. This is the date until which it guarantees the potency and sterility of the drug in its original container.
USP <797>, the standard for sterile compounding, has very specific guidelines for BUDs. The BUD is a conservative estimate of the time to use a compounded sterile preparation.
It considers several factors including the stability of the components, how the product is stored and the risk of microbial contamination during the compounding process. For low-risk, simple sterile products stored in the range of 2°C to 8°C, the default BUD is 28-45 days.
The FDA states, “the expiration date is determined by the manufacturer and is not the same as the beyond-use date assigned by a compounder.” And pharmacy practice standards also encourage using BUDs, such as the American Pharmacists Association (APhA).
If an injectable compounded product is used after the BUD, there are two major concerns:
- The concentration of the active ingredient may be less than labeled.
- The preservative (if used) may be ineffective to inhibit microbial growth and cause sterility problems
Patient Implication: A patient may be dispensed a 10 mL bottle of semaglutide with a BUD of 30 days. They will have 12 weeks of supply if they are starting at a low dose (e.g., 0.25 mg/week). The patient can use this for many months, if it’s similar to a manufacturer’s vial. At weeks 9-12 they are likely to be injecting a diluted solution (less active) so less effect.
4. Human Factors of Dosing Confusion and Syringe Errors
The step up from a pre-filled, dose-calibrated manufacturer’s pen to a multidose vial and insulin syringe is huge, and therefore more prone to error. FDA-approved pens are designed with human factors in mind; you dial and click to get a dose.
Compounding insulin requires the patient to do the dosing. The prescription might say, “Inject 0.25 mg (10 units) weekly”. This requires the patient to understand:
- What strength of insulin they are using (e.g. 2.5 mg/mL or 5 mg/mL).
- That the “units” on an insulin syringe represent volume, rather than weight (mg).
- How to convert the insulin weight (mg) into volume (mL) for their insulin.
The Institute for Safe Medication Practices (ISMP) has long classified insulin and other injectable medications that are measured in syringe “units” as a high risk for medication errors. Confusion with “units” and “mLs”, wrong syringe (e.g. U-100 vs. U-40) and faulty reading of syringe barrel markings are frequent potentially lethal errors (ISMP Injection Safety.
Medicine errors reports from the FDA also indicate that unclear medication labels, ambiguous abbreviations and complex dose calculations are also common causes of medication errors in injectable medications
This risk is highlighted in the literature. One study in the Journal of Patient Safety has demonstrated that despite being given clear instructions, many patients make errors in calculating doses of injectable medications in vials. These are almost always under-doses, rather than over-doses.
Patient Implication: A patient with a 5 mg/mL vial, who draws 10 units (0.1 mL) thinking this is 0.25 mg, has drawn 0.5 mg (twice the dose). A patient with a 2.5 mg/mL vial, who draws 5 units (0.05 mL) thinking it is 0.25 mg, has drawn 0.125 mg (a half dose). This certainly would be interpreted as being ineffective.
5. The Black Market of “Research Chemicals” and Counterfeits
A black market has sprung up alongside the pharmaceutical industry, driven by the profits of semaglutide. A simple Google search reveals numerous sellers of vials of “semaglutide” or “semaglutide sodium” – some also marked as “For Research Use Only” or “Not for Human Consumption”.
FDA’s Guidelines mention strict controls for RUO products. The FDA’s labeling guide states that RUO products are only to be used for animal studies in a lab. They clearly do not have to meet the safety, purity and potency standards of human drugs. Their labels are regulatory disclaimers, not a problem.
The consequences of sourcing medicines from this “market” are grave:
- Unknown Purity and Content: The product in the vial is not checked. It may contain less semaglutide, no semaglutide or contaminants (metals, bacterial endotoxins) as a result of non-sterile manufacturing.
- Sterility Failures: These products are not made in USP <797> compliant cleanrooms. Infection or abscesses may be caused when injected.
- Counterfeits: The FDA Drugs Guidance is regularly warning about counterfeit drugs that may appear to be the real product but contain the wrong active ingredients, wrong dose or contaminants.
Patient Implication: Online purchase of “research chemical” semaglutide is an uncontrolled experimentation for a patient. A chance of receiving a pure potent and safe drug is a significant cause of therapy failure and side effects.
6. The Unproven “Boosting” with Additive Effects of B12, B6 and L-Carnitine
We are living in a competitive society and some compounding pharmacists advertise “better” semaglutide with added ingredients such as vitamin B12 (cyanocobalamin or methylcobalamin), vitamin B6 (pyridoxine) or an amino acid derivate L-carnitine. This is claimed to boost energy, reduce side effects (nausea) and aid fat burning. But it increases the complexity and risk.
The United States Pharmacopoeia’s (USP) chapter <795> describes the guidelines of the United States Pharmacists for non-sterile compounding (and by inference, sterile admixtures) which states that if ingredients are mixed, such practice is justified by the evidence of compatibility and stability. Just because two substances can be injected separately is no guarantee that they will be stable in the same vial for weeks on end.
The Office of Dietary Supplements of National Institutes of Health gives fact sheets about certain nutrients, including vitamin B12, in which they clarify that while generally considered safe, there are certain concerns regarding the dosage, form, and stability of the injectable version of vitamin B12 as compared to oral supplements.
It has been noted in the compounding literature, specifically theInternational Journal of Pharmaceutical Compounding , that certain additives in a formulation might actually affect the stability of the primary drug in the mixture. The pH, ionic strength, and perhaps even chemical reactions within the formulation might cause faster degradation of the semaglutide peptide, thereby reducing its BUD still further.
Implication for Patient: Additives are probably safe when used in typical amounts but might actually cause changes in the molecular structure of the semaglutide peptide, thus making it denature faster inside the vial. The patient might then be spending more money on an altered drug with a lesser effectiveness and potency than the normal semaglutide.
List of Common “Compounded Semaglutide” Telemedicine Brands
| Company | About | Semaglutide Price | Compounding Pharmacy & Location | Certifications / Notes |
| Hims & Hers | A telehealth provider of weight loss programs has offered compounded semaglutide in “personalized” doses (subject to change). | About $199/month (varies with subscription); media reports $165/month. | Uses its own telehealth network; compounded meds sourced from licensed U.S. pharmacies (e.g., BPI Labs previously). | Operates with U.S. telehealth licensure; has faced FDA warning letters about marketing claims. |
| Ro | Online telehealth company that specialises in telehealth; prescribes FDA-approved GLP-1 medications (such as Ozempic and Wegovy) through insurance/cash pay. (Ro) | Cost for medication varies – membership costs ~$45 first month, then ~$145/month + medication. Wegovy cash pay (some doses) ~$199-$349/mo. (Ro) | Partners with established pharmacies (e.g., LillyDirect®, NovoCare®) for FDA-approved meds. (Ro) | LegitScript/Telehealth regulated; focuses on FDA-approved meds. (no compounding pharmacy data for semaglutide compounding) |
| Henry Meds | Online telehealth service providing GLP-1, hormone replacement therapy and weight loss. | From $197 per month (advertised) | Licensed U.S. mail-order pharmacies handle fulfillment (exact pharmacy varies by patient). | Operates under U.S. telehealth and mail pharmacy regulations (no specific external certification publicly listed). |
| Orderly Meds | Online/telehealth program marketing compounded GLP-1 injections for low “starter” costs. (OrderlyMeds) | Promotes a “simple start” for 3 months, $249 (promotional claim). | Telehealth + U.S. compounding pharmacy partners (not always disclosed on site). | Standard U.S. telehealth oversight; specific certifications not publicly listed. (OrderlyMeds) |
| Mochi Health | Subscription service with medication add-on; publicly advertises compounded semaglutide (joinmochi.com) | Membership $79/mo plus $99/mo for compounded semaglutide (as displayed). | Delivered by partner pharmacies (U.S.). | Standard U.S. telehealth regulations; individual pharmacy credentials vary by partner. |
| Brello Health | Telehealth/partner providers offering access to compounded GLP-1 drugs. (BrelloHealth) | The product page states $399 for the plan that includes compounded semaglutide. | Meds dispensed through partner licensed U.S. pharmacies (transitioning facilities). | Operates within U.S. telehealth; pharmacy certifications dependent on partners. |
| Levity | Telehealth weight loss program + compounded semaglutide (their product pages claim it has addins such as B12/glycine). | Product page shows $225 every 4 weeks (first/subsequent order pricing may vary by promo). | Uses in-house (or partnered) compounding/dispensing facilities; operates nationwide. | U.S. telehealth regulated; pharmacy may hold standard U.S. pharmacy licenses (no public third-party certifications shown). |
| Weight Watchers / WW | Telehealth + weight program. Advertises semaglutide; has moved away from compounded semaglutide to partnerships with brand names (e.g. Wegovy). | WW clinic page shows starting at $199/mo for self-pay options (often branded), and Reuters reported a $299 Wegovy cash price via Novo partnership for a period. | Compounding pharmacy: N/A (reported stop of compounded semaglutide). | N/A
|
| Noom Med | Weight-loss program + medications; Reuters notes Noom offered compounded semaglutide at lower/personalized doses and intro prices.
.. |
Noom Med page shows starting at $149; Reuters reported $119 intro month, then $199/mo after (for a max 0.6 mg dose in that report). | Not publicly disclosed | Not publicly disclosed
|
| IVIM Health | Telehealth weight-loss program that prescribes compounded semaglutide.
|
Public pages show starting as low as $149/mo; separate help article shows compounded semaglutide “starting at $75/mo” plus membership (pricing depends on plan structure). | Not named publicly, but a peer-reviewed article states IVIM uses 503A or 503B compounding pharmacies in the U.S. | 503A or 503B partner pharmacies (per published article).
|
| PlushCare | Virtual clinic; provides access to semaglutide and has a partner pharmacy cash-pay option for compounded semaglutide (if permitted). | $149/mo (0.25 mg) and $299/mo (0.5–2.4 mg) cash-pay (eligible states). | Not publicly disclosed (describes “preferred state-licensed sterile compounding pharmacy partner”). | States partner is state-licensed sterile compounding and compliant with federal/state law; no pharmacy name given, so no PCAB verification publicly. |
| Calibrate | Metabolic program; states they do not prescribe compounded meds. | N/A for compounded semaglutide. | N/A | N/A |
| Eden (tryeden.com) | Online prescribing service; mentions compounded drugs from state-licensed pharmacies. | (Pricing varies by offer; page shown emphasizes dispensing model more than a single national price.) | Not publicly disclosed (states “state-licensed pharmacies”). | “State-licensed pharmacies” (no named pharmacy → can’t verify PCAB/503A/503B publicly). |
| Sunlight (sunlight.com) | Telehealth weight-loss program; says it includes GLP-1 medications (semaglutide/tirzepatide) + visits + shipping.
|
Price not visible in the snippet I captured (site emphasizes what’s included). | Not publicly disclosed | Not publicly disclosed |
| New Self | Online weight-loss & telehealth clinic offering semaglutide and tirzepatide plans; advertises licensed pharmacy partners. | Promotes competitive pricing like ~$199 semaglutide membership (flat price across doses). | Partner pharmacies (e.g., Beluga Health P.A. and LegitScript-aligned partners per comparison site). | Mention of LegitScript-aligned pharmacy partners and 503A compounding context via third-party comparison. |
| Refills (Refills.com) | Online telehealth & pharmacy platform offering prescription weight-loss and general pharmacy services (non-specific GLP-1/semaglutide details). | Not publicly listed for semaglutide pricing. | Network of licensed healthcare professionals + online pharmacy services. | Licensed U.S. pharmacy network; specifics not provided. |

Conclusion: 3-Step Quality Audit
If you are not getting desired results from compounded semaglutide, don’t assume you are a “non-responder”. Thoroughly investigate the drug itself. Here’s your 3-step quality audit:
Step 1: Examine the Product
Clarity: When you get a new vial, check. It should be clear, colorless and without any particles, cloudiness or haze. Semaglutide base in sterile water or bacteriostatic water is clear. Any visual anomaly is not good, don’t inject it.
Labeling: Pharmacy label must include:
- The name of the patient and prescriber.
- The concentration (i.e., “Semaglutide 2.5 mg/mL”).
- The Beyond-Use Date (BUD) is not an expiry date.
- Storage information (“Store at 2°C-8°C”).
- Lot number for traceability.
Step 2: Check the Credentials
Pharmacy Legitimacy: Is the pharmacy a 503A compounding pharmacy (compounding individual prescriptions) or 503B outsourcing facility (compounding larger amounts of sterile products)? You should check this.
- Verify an internet pharmacy through the National Association of Boards of Pharmacy (NABP) Verified Internet Pharmacy Practice Sites (VIPPS) program or their “.pharmacy” domain name verification program.
- Check with your state’s Board of Pharmacy to ensure the pharmacy is licensed.
Don’t Use Gray Markets: If you are referred to a website that sells “research peptides” or if the transaction is covert, it’s illegal and dangerous. Prescriptions are filled at legitimate pharmacies not by online vendors.
Step 3: Double Check the Protocol
Consult the Pharmacist: Book in an “in-person” (phone or video) consultation with the compounding pharmacist before your first injection. Don’t just rely on written instructions. Ask them to explain:
- “How many milligrams (mg) or units per milliliter (mL) are in my vial?”
- “How many milliliters (mL) or units on my syringe, depending on the syringe, should I draw for my dose of X mg?”
- “Can you verify for me while I have the vial and syringe at hand?”
Leverage Professional Resources: Websites of professional organizations such as the American Society of Health-System Pharmacists (ASHP) provide information on drug shortages and sterile preparation, highlighting the need for pharmacy professional oversight when preparing doses and confirming medication labels.
References:
-
- https://www.ashp.org/drug-shortages/current-shortages?loginreturnUrl=SSOCheckOnly
- https://safe.pharmacy/
- https://ijpc.com/Abstracts/Abstract.cfm?ABS=4697
- https://ods.od.nih.gov/factsheets/VitaminB12-HealthProfessional/
- https://www.usp.org/compounding/general-chapter-795
- https://www.fda.gov/drugs/guidance-compliance-regulatory-information/guidances-drugs
- https://www.fda.gov/regulatory-information/search-fda-guidance-documents/distribution-in-vitro-diagnostic-products-labeled-research-use-only-or-investigational-use-only
- https://journals.lww.com/journalpatientsafety/Fulltext/2018/06000/Medication_Dosing_Errors.6.aspx
- https://link.springer.com/article/10.1007/s40278-024-54314-7
- https://www.usp.org/compounding/general-chapter-797
- https://onlinelibrary.wiley.com/doi/10.1002/psc.70039
- https://extranet.who.int/prequal/sites/default/files/document_files/WHO_TRS_961_eng.pdf
- https://www.pharmagmp.in/how-fda-gmp-regulations-influence-drug-storage-and-handling/#google_vignette
- https://www.uspharmacist.com/article/fda-warns-about-counterfeit-improperly-compounded-semaglutide-products
- https://www.fda.gov/drugs/human-drug-compounding/compounding-when-drugs-are-fdas-drug-shortages-list
- https://home.ecri.org/pages/ismp
Read this Next
How Do You Store Semaglutide Medication?
Semaglutide is the active ingredient in drugs sold under the brand name of Ozempic, Wegovy, and Rybelsus that helps...
Semaglutide: How it Works, Pros & Cons, Estimated Weight Loss
You’ve heard of Semaglutide. Maybe without actually knowing it. This weight loss medication has taken on a life of...